 
Note: when the addition of the Greek prefix places two vowels adjacent to one another, the "a" (or the "o") at the end of the Greek prefix is usually dropped e.g., "nonaoxide" would be written as "nonoxide", and "monooxide" would be written as "monoxide". SF 6 Sulfur Hexafluoride (Note that there is only one Sulfur but no Mono prefix) "Mono" is not used to name the first element SF 6 Sulfur Hexafluoride (There are 6 Fluorines so Hexa is used as the prefix)Ĥ. Prefixes are used to denote the number of atoms SF 6 Sulfur Hexa fluoride (Fluorine becomes Fluoride)ģ. Second element is named using the suffix "-ide" The first element is named first, using the elements name:Ģ. Nomenclature is the fancy way of saying the rules for naming.ġ. Now that you can form covalent compounds we need to go over how to name these compounds. If that same hydrogen bonded to Chlorine, the hydrogen would get the two electrons it needs to be complete and the chlorine which has 7 valence electrons would get the one more to fulfil its octet. For instance, looking at hydrogen we know that it is in Group I and thus has 1 valence electron, if it bound itself to another hydrogen they could share the two electrons between them and both be "happy". Now that you know the number of valence electrons and the rules you can start making molecules. They have extra space to allow for the extra electrons. Elements on the 3rd Period (3rd row) of the periodic table and below can actually have more than 8 electrons around them.Hydrogen (H) only requires 2 electrons to have a full valence shell since it only needs to be like Helium (He).There are several exceptions to the octet rule however:.The Noble Gases are like the Michael Jordan's of the Chemistry world and every one of them wants to "be like Mike". This means that they want to acquire the electronic configuration of the noble gas of their row. The Octet Rule: The atoms that participate in covalent bonding share electrons in a way that enables them to acquire a stable electron configuration, or full valence shell.There are a couple of rules to follow however as you build: Once you know how many valence electrons an atom has, you can start to build molecules. For example, Boron (represented as B in the periodic table) is in Group 3A and has 3 valence electrons Carbon (represented as C) is in Group 4A and has 4 valence electrons. Conveniently, the Group Number at the top of each column in the periodic table also gives the number of valence electrons. In the periodic table below, you can see diagrams of each element that shows how many valence electrons it possesses. The bonding electrons are called the VALENCE electrons and they are the electrons that are found in the outermost shell of the atom. What we did not discuss previously is which electrons in the atom are involved in bonding. Covalent BondsĪs we mentioned before, the electrons in an atom are what is responsible for forming bonds. Combustion of alkanes and other hydrocarbons.© 2008 WGBH Educational Foundation. 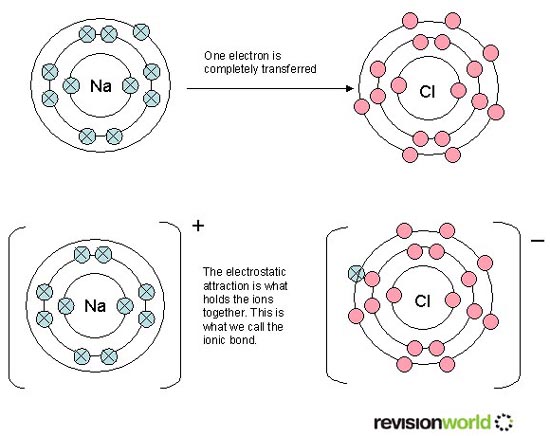
Exothermic and endothermic reactions (and changes of state). Chemical reactions can result in a change in temperature.5.4 Exothermic and Endothermic Reactions.Characteristics of covalent substances.Single, double and triple covalent bonds. Covalent bonding as the sharing of pairs of electrons.(d) the accepted structural model for giant ionic structures in explaining the physical properties of ionic compounds.(c) electronic structure in explaining how ionic bonding takes place (and how this is represented using dot and cross diagrams).Unit 2: CHEMICAL BONDING, APPLICATION OF CHEMICAL REACTIONS and ORGANIC CHEMISTRY.(a) ionic bonding in terms of ion formation and the interaction between positive and negative ions in an ionic crystal.Unit 1: THE LANGUAGE OF CHEMISTRY, STRUCTURE OF MATTER AND SIMPLE REACTIONS.NaCl, MgCl₂) and relevant energy terms for: the construction of Born–Haber cycles and related calculations b) use of the lattice enthalpy of a simple ionic solid (e.g.Module 5: Physical chemistry and transition elements.a) ionic bonding as electrostatic attraction between positive and negative ions, and the construction of 'dot-and-cross' diagrams.be able to construct Born-Haber cycles and carry out related calculations know that ionic bonding is the strong electrostatic attraction between oppositely charged ions Born–Haber cycles are used to calculate lattice enthalpies using the following data: enthalpy of formation, ionisation energy, enthalpy of atomisation, bond enthalpy, electron affinity.Ionic bonding involves electrostatic attraction between oppositely charged ions in a lattice.RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
